 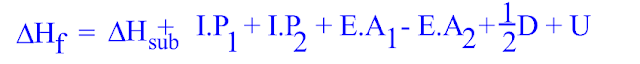  
The amount of energy released when an electron attaches to a neutral atom or molecule in the gaseous state to form an anion is defined as an atom's or molecule's electron affinity. The minimum energy required to remove the most loosely bound electron from an isolated gaseous atom, positive ion, or molecule is referred to as ionisation energy. In the Born Haber Cycle, Hess' Law is effectively applied to an ionic solid.īefore the Born-Haber Cycle can be used to calculate the lattice energy of an ionic solid, the following concepts must be understood: Hess' Law states that the overall change in energy of a process can be calculated by breaking it down into phases and then summing the energy changes of each phase. Those forces are only completely broken when the ions are present as gaseous ions, spread so far apart that the attraction between them is negligible. The stronger the forces, the greater the lattice enthalpy. Lattice enthalpy measures the strength of the forces between the ions in an ionic solid. In order to calculate lattice energy (or, more precisely, enthalpy), which cannot be measured directly, Born-Haber cycles are primarily used. The cycle is concerned with how a metal-typically a Group I or Group II element-reacts with a halogen or another non-metallic element, like oxygen, to form an ionic compound. It was named after Max Born and Fritz Haber, two German scientists who invented it in 1919Additionally, it was independently developed by Kasimir Fajans and simultaneously published in the same journal's issue. The Born-Haber cycle is a method for calculating reaction energies. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
